Read the following text and answer the questions that are posed below:
The energy of stars, and therefore of the Sun, comes from nuclear reactions that take place in their interior.
In 1925, the astronomer Cecilia Payne demonstrated that the predominant components of a star are primarily 11H and 42He in smaller proportions, which means that inside the star there is a true factory of atoms.
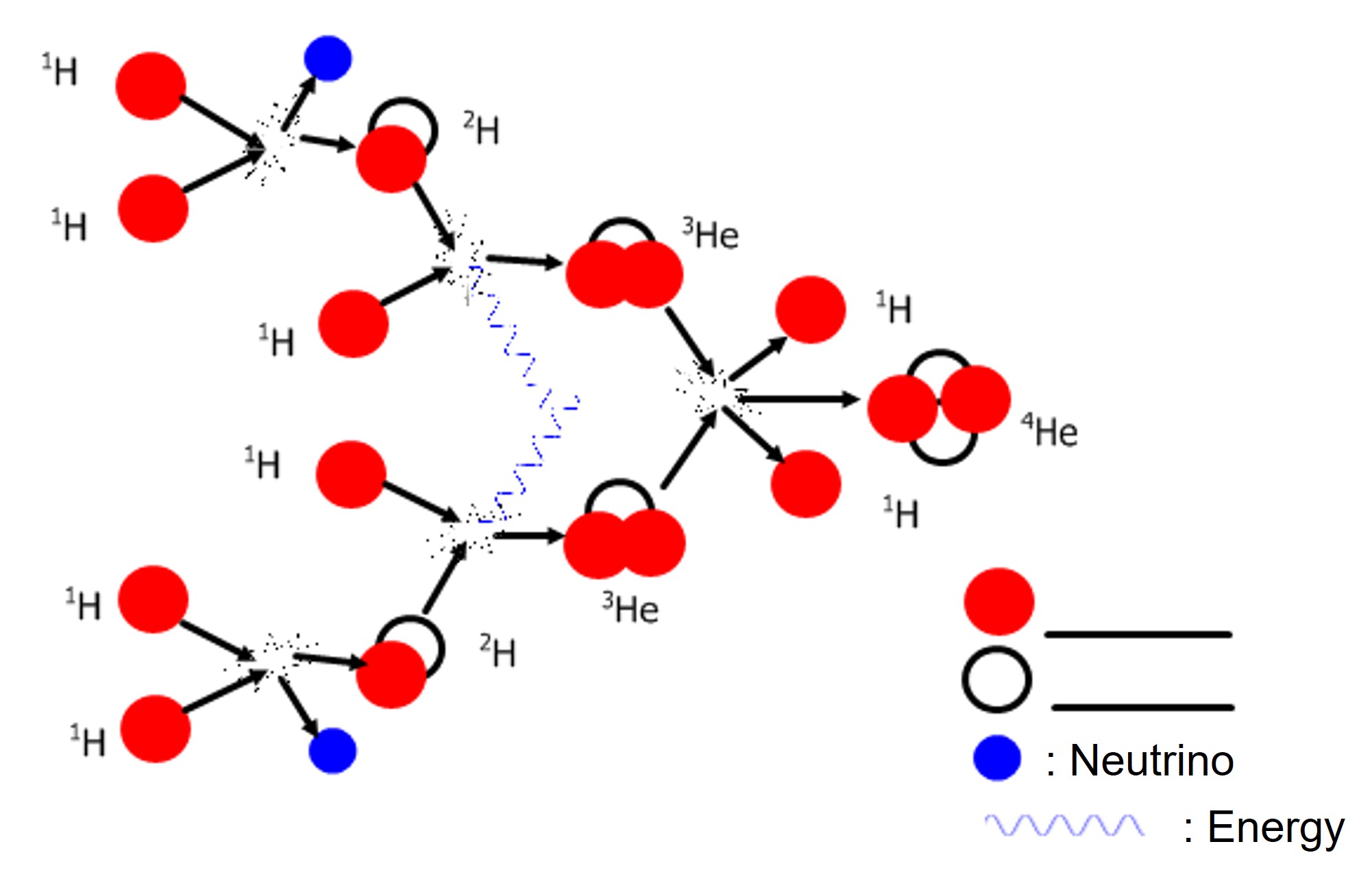
The nucleus of a hydrogen atom consists of a single particle, the proton. The fusion of two hydrogen nuclei results in a deuterium nucleus, 21H , which is an isotope of hydrogen. When deuterium fuses with another hydrogen nucleus, it forms an isotope of helium with mass number 3. Then two helium-3 nuclei collide and form an ordinary helium nucleus 42He.
After the formation of helium, the process of nuclear transformations continues with the successive creation of other elements heavier than helium, such as: 74Be, 73Li and continues up to carbon, nitrogen, oxygen, etc.
(Adapted from the text proposed as a teacher's resource from Editorial Santillana -Proyecto La Casa del Saber, for 3rd ESO).
1) What does the phrase "inside a star, there is a true factory of atoms" mean?
2) In the diagram, indicate the name of the particles represented as:

3) The text describes the following nuclear reaction: “ When deuterium fuses with another hydrogen nucleus, it forms an isotope of helium of mass number 3”.Circle the graphical representation where the process described in the previous sentence takes place.
5) Look at the nucleus in the figure below and indicate: to which element it corresponds, what is its mass and atomic number and write the symbol of the isotope.

6) Explain what is the composition of the nuclei represented by the symbols 11H and 42He.
7) Are atoms 74Be and 73Li isotopes and why?
8) Indicate which of the following symbols are wrong: 42Li , 32H, 31H, 83Li.
9) How are the reactions described in the text called?
