The photoelectric effect occurs when we irradiate a metal with light in such a way that it manages to remove electrons from its surface, as shown in the image.
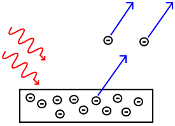
In order to remove the electrons, it is necessary for the light to have a minimum frequency (a colour), called the cutoff frequency, which is what manages to remove the first electrons. As we increase this frequency, the electrons have greater energy and, therefore, they leave with greater speed.

Mileva Marić (1875-1948), a brilliant 20th century Serbian mathematician, studied this phenomenon, although it was her husband, Albert Einstein, who won the N obel Prize for this work.
1.- The following graph represents the energy (OY axis) of the removed electrons, as a function of the frequency (OX axis) of the light that reaches the metal.
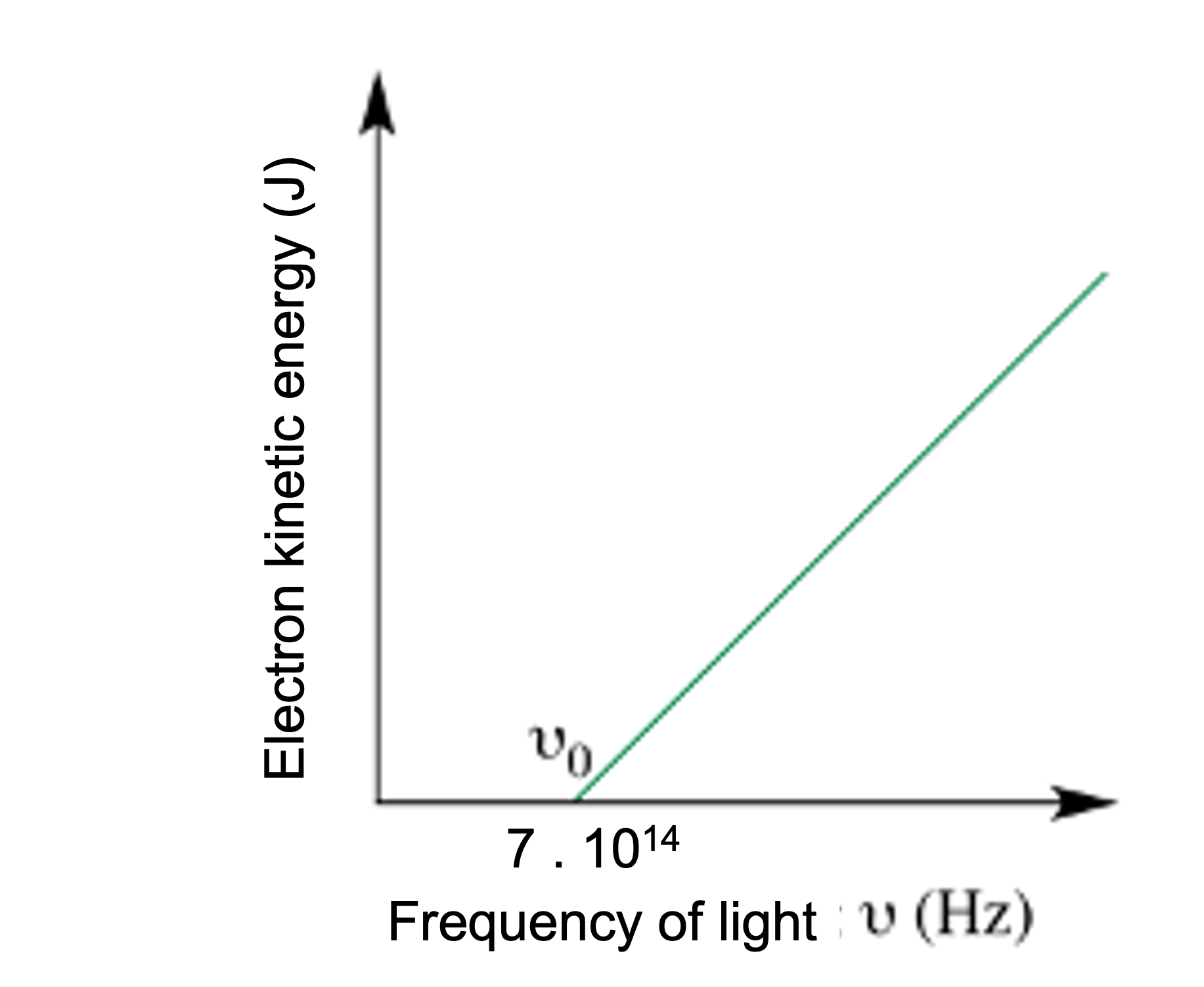
a) What kind of function is it?
b) Does the function increase or decrease?
c) At what value of frequency is the first electron ripped off? With what energy is that electron released?
d) For which values of the frequency can we not remove electrons?
2.- Each metal has a different cutoff frequency, that is, the first electrons start to come out at different frequencies. To identify the type of metal with which you want to work, light is irradiated by changing the frequency, and when we detect the first electron, we know what metal it is. Below, we can find a graph showing the energy function of the electrons removed by different elements (rubidium, sodium, potassium, barium, and calcium), as a function of the radiated frequency.
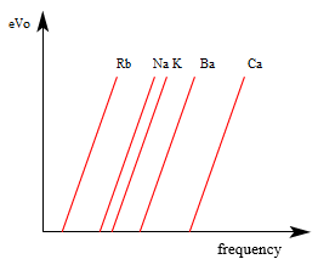
a) Which metal has the lowest cutoff frequency? Which one has the largest?
b) What is the relative position of the graph of these functions?
